Neon Isotopes Illustration Physics Sciences Backdrops Stock Illustration - Illustration of physics, planetary: 217364251

How many grams of neon will have the same number of atoms as 4 grams of calcium ? (Atomic masses : ` - YouTube

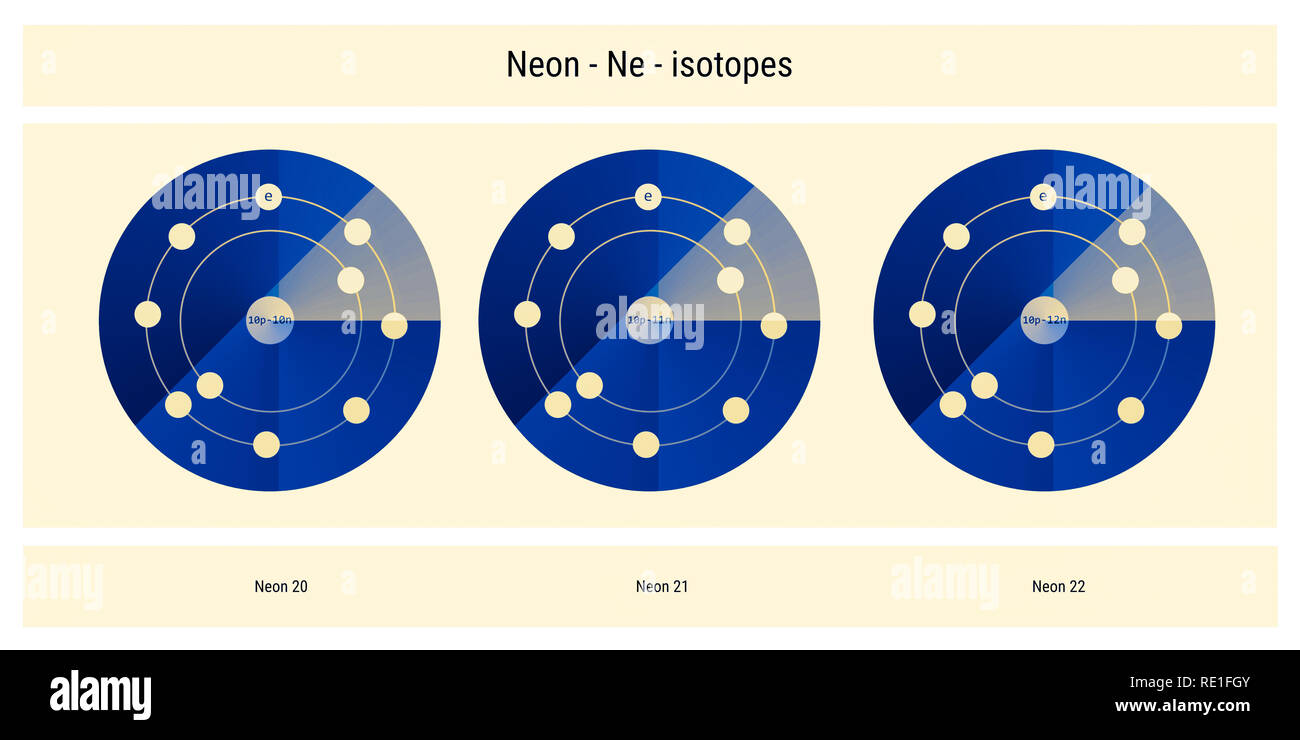
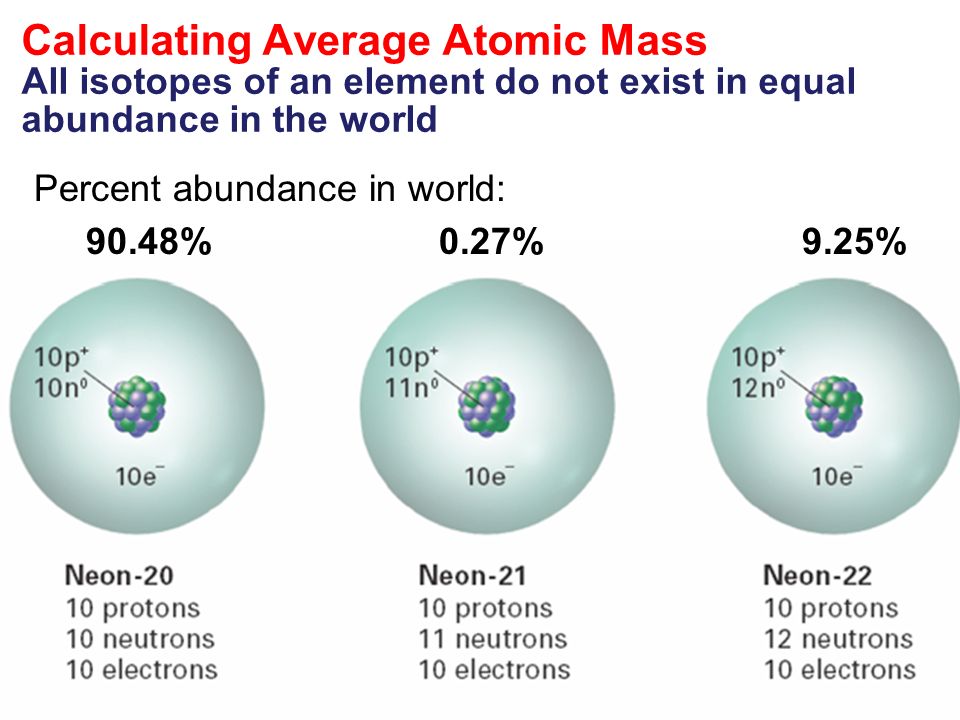
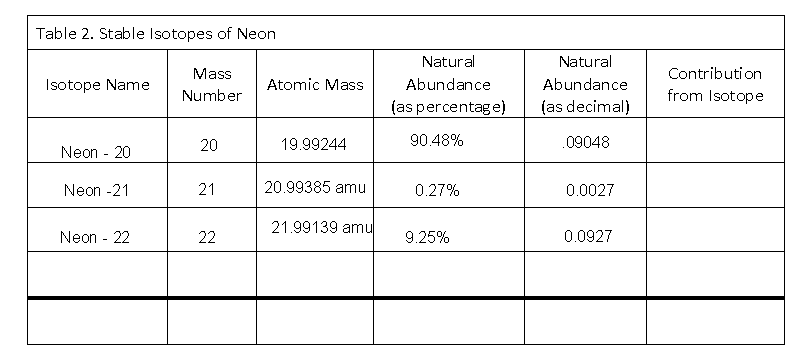
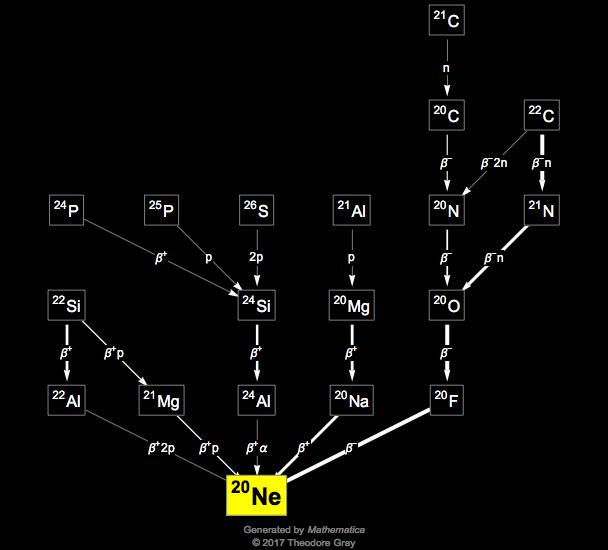
The three stable isotopes of neon Ne^20, Ne^21 and Ne^22 have respective abundances of 90.51% , 0.27 % and 9.22% . The atomic masses of the three isotopes are 19.99 u, 20.99